Peer Reviewed
Non-Selective β-Blocker Use in the Treatment of Stewart-Treves Syndrome
AFFILIATIONS:
1Lake Erie College of Osteopathic Medicine, Erie, Pennsylvania
2Department of Dermatology, Milton S. Hershey Medical Center, Hershey, Pennsylvania
3Department of Orthopaedics and Rehabilitation and Medicine, Milton S. Hershey Medical Center, Hershey, Pennsylvania
4Department of Hematology and Oncology, Milton S. Hershey Medical Center, Hershey, Pennsylvania
CITATION:
Axcell K, Kim, Y, Fox EJ, Drabick JJ, Anderson BE. Non-selective β-blocker use in the treatment of Stewart-Treves syndrome. Consultant. 2023;63(3):e9 doi:10.25270/con.2023.01.000006
Received July 7, 2022. Accepted November 21, 2022. Published online January 18, 2023.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Kent Axcell, BS, 6642 Traquair Place, Dublin, OH, 43016 (AxcellK@Gmail.com)
Introduction. A 60-year-old man presented to the emergency room with a left upper extremity angiosarcoma secondary to chronic lymphedema of the left upper extremity (Figure 1).
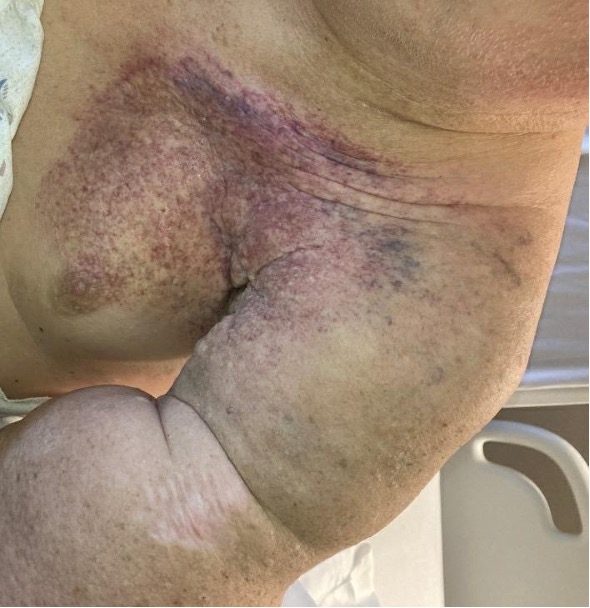
Figure 1. Left upper extremity chronic lymphedema and angiosarcoma.
Patient history. The patient’s history included a malignant melanoma of the left arm, leading to a wide local surgical excision and sentinel lymph node biopsy in 2014. The patient’s sentinel lymph node biopsy was positive for metastasis, but the patient denied adjuvant interferon therapy at that time. In 2016, he developed a 10 x 7 x 7 cm nodal recurrence of the left axilla and underwent lymph node resection. However, after the lesion developed again, the patient underwent radiotherapy and systemic immunotherapy with intravenous ipilimumab 3 mg/kg and nivolumab 1 mg/kg, followed by nivolumab 240 mg every 2 weeks with notable nodal regression and lack of disease progression. His case was complicated by severe lymphedema of the left upper extremity. In 2020, he noted a protuberant, fungating mass of the left upper posterior arm that was consistent with angiosarcoma (Figure 2).
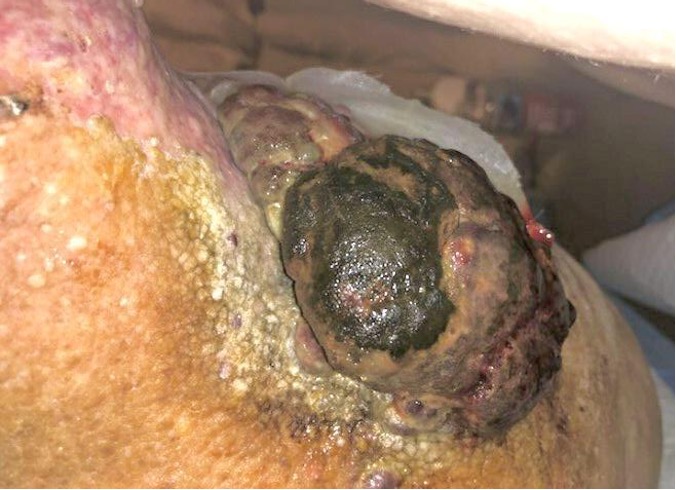
Figure 2. A protuberant, fungating angiosarcoma tumor of the left upper posterior arm.
Diagnostic testing. The patient underwent computed tomography (CT) then magnetic resonance imaging (MRI) of the chest and upper extremity, which showed post-operative and post-radiation changes without a clear mass.
Treatment and management. The patient had a palliative amputation of the left upper extremity; however, he also had dermal satellite lesions at the chest wall, which rendered his overall disease burden incurable. Caris Next-Generation Sequencing identified no actionable mutations and the patients’ total mutational burden was low, making immunotherapy success unlikely. He received weekly paclitaxel 80 mg/m2 for the satellite lesions with noted tumor regression. Propranolol 2 mg/kg was also added for its potential anti-angiosarcoma effects. Although the addition of propranolol appeared to have stopped the large disease burden from recurring after maximum response to chemotherapy, he no longer responded to paclitaxel and the chest wall lesions continued to progress.
Patient outcome. The patient started doxorubicin with a plan for the patient to undergo palliative radiation. However, the patient died in November 2021.
Discussion. Stewart-Treves syndrome is a rare manifestation of cutaneous angiosarcoma secondary to chronic lymphedema. A malignancy of vascular or lymphatic endothelial cell origin, cutaneous angiosarcoma (cAS) can arise anywhere on the body, most commonly on the head or neck of older White men.1 Unfortunately, cAS carries a poor prognosis with a mean survival of about 1 year and a 5-year survival rate of 33.5%.2 The pathogenesis of cAS is due to the proliferation of endothelial cells with invasion of surrounding tissue.1 Other causes of cAS include radiation or secondary to a foreign body.1,3 The standard treatment for localized cAS is surgery followed by postoperative radiotherapy. Paclitaxel is the current standard of care for advanced disease.2 Propranolol and other non-selective beta-blockers, such as timolol, have been used as the treatment of choice in high-risk juvenile hemangiomas and adjunct agents in the treatment of Kaposi sarcoma.4,5 Indeed, the use of propranolol for cAS treatment has been encouraging, with evidence showing a synergistic effect with select chemotherapeutic agents, leading to improved patient outcomes.6
The blockade of the beta-adrenergic receptor on vascular endothelial cells has demonstrated involvement in the oncogenic pathway by obstructing the action of epinephrine and norepinephrine on pro-angiogenic mediators such as HIF-1α, vascular endothelial growth factor (VEGF), and interleukin-6 (IL-6). β-adrenergic activation in the vascular tumor microenvironment results in a decrease in cytotoxic T-cell invasion and an increase in tumor-associated macrophage activation. Adrenergic signaling is believed to be the driving force behind endothelial cell migration and proliferation, highlighting the hypothesized mechanism β-blockade slowing tumor growth and metastasis.7
cAS is extremely locally invasive and readily metastasizes. Asymptomatic red papules or nodules that coalesce into large, fleshy masses of red-tan to gray-white tissue with indistinct margins, hemorrhage, and central necrosis are typically seen on examination.3 Histopathological examination shows irregularly shaped anastomosing vascular channels with nuclear atypia, staining positive for CD31, CD43, VEGF, electroretinogram, and/or factor VIII with high expression of β-adrenergic receptors. This suggests a major role for the implementation of β-blockers in the treatment course. 8 The current standard of care for localized angiosarcoma remains surgery followed by radiotherapy. Advanced disease, however, is generally treated with paclitaxel, a microtubule targeting chemotherapeutic agent. Agents such as bevacizumab, eribulin mesylate, trabectedin, and PD-L1 targeting agents have been trialed with unclear results.2
The efficacy of propranolol in the treatment of other vascular lesions such as infantile hemangiomas (IH) is strongly supported. It has been shown to inhibit growth and induce IH regression leading to FDA approval in 2014 for IH requiring systemic therapy. Hypothesized mechanisms include vasoconstriction, inhibition of VEGF, basic fibroblast growth factor, and triggering apoptosis.5 Propranolol and other non-selective beta-blockers have been utilized as an adjunct treatment in vascular malignancies cAS and Kaposi sarcoma with beneficial outcomes.9 Propranolol has shown a synergistic effect, specifically in combination with chemotherapeutic agent vinblastine in the treatment of cutaneous angiosarcoma.2 In a 2015 study, Chow and colleagues10 reported that monotherapy with the β-blocker propranolol resulted in a 34% reduction in the proliferative index of a β-adrenergic-positive cAS involving over 80% of the scalp, face, and cheek. The lesion was subsequently treated with propranolol, paclitaxel, and radiotherapy leading to further regression and no metastasis.10 We have yet to see strong recommendations for the use of propranolol due to the lack of robust randomized, controlled studies. In the case described, the use of propranolol led to a substantial clinical improvement of the large disease burden. Given the molecular evidence present regarding adrenergic involvement in vascular tumor progression and evidence of efficacy in infantile hemangioma, further investigation into standardizing the use of propranolol in the treatment of cutaneous angiosarcoma is needed.
- Young RJ, Brown NJ, Reed MW, Hughes D, Woll PJ. Angiosarcoma. Lancet Oncol. 2010;11(10):983-991. doi:10.1016/s1470-2045(10)70023-1.
- Ishida Y, Otsuka A, Kabashima K. Cutaneous angiosarcoma: update on biology and latest treatment. Curr Opin Oncol. 2018;30(2):107-112. doi:10.1097/cco.0000000000000427.
- Cotran RS, Kumar V, Robbins SL. Robbins and Cotran pathologic basis of disease. 10th ed. Elsevier; 2005.
- McAllister SC, Hanson RS, Manion RD. Propranolol decreases proliferation of endothelial cells transformed by Kaposi's sarcoma-associated herpesvirus and induces lytic viral gene expression. Virol J. 2015;89(21):11144-11149. doi:10.1128/jvi.01569-15.
- Metry DW, Levy ML, Corona R. Infantile hemangiomas: management. UpToDate. Updated November 7, 2022. Accessed August 25, 2021. Available at: https://www.uptodate.com/contents/infantile-hemangiomas-management?search=angiosarcoma+propranolol&topicRef=7729&source=see_link.
- Pasquier E, André N, Street J, et al. Effective management of advanced angiosarcoma by the synergistic combination of propranolol and vinblastine-based metronomic chemotherapy: a bench to bedside study. EBioMedicine. 2016;6:87-95. doi:10.1016/j.ebiom.2016.02.026.
- Wagner M, Cranmer L, Loggers E, Pollack S. Propranolol for the treatment of vascular sarcomas. J Exp Pharmacol. 2018;10:51-58. doi:10.2147/jep.s146211.
- Liu SS, Bellamkonda V, Phung TL. Angiosarcoma. Published March 30, 2021. Accessed July 16, 2021. Available at: https://www.pathologyoutlines.com/topic/softtissueangiosarcoma.html.
- Meseguer-Yebra C, Cardeñoso-Álvarez ME, Bordel-Gómez MT, Fraile-Alonso MC, Pérez-Losada ME, Sánchez-Estella J. Successful treatment of classic Kaposi sarcoma with TOPICAL timolol: report of two cases. Br J Dermatol. 2015;173(3):860-862. doi:10.1111/bjd.13746.
- Chow W, Amaya CN, Rains S, Chow M, Dickerson EB, Bryan BA. Growth attenuation of cutaneous angiosarcoma with propranolol-mediated β-blockade. JAMA Dermatol. 2015;151(11):1226. doi:10.1001/jamadermatol.2015.2554.


